Cho 15,2g hỗn hợp gồm rượu etylic và axit axetic tác dụng vừa đủ với 400ml dung dịch NaOH 0,5M. Tính thành phần phần trăm khối lượng của mỗi chất trong hỗn hợp ban đầu
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


a, Gọi \(\left\{{}\begin{matrix}n_{C_2H_5OH}=a\left(mol\right)\\n_{CH_3COOH}=b\left(mol\right)\end{matrix}\right.\)
\(n_{H_2}=\dfrac{11,2}{22,4}=0,5\left(mol\right)\)
PTHH:
2C2H5OH + 2Na ---> 2C2H5ONa + H2
a---------------------------------------->0,5a
2CH3COOH + 2Na ---> 2CH3COONa + H2
b------------------------------------------------>0,5b
=> hệ pt \(\left\{{}\begin{matrix}46a+60b=48,8\\0,5a+0,5b=0,5\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}a=0,8\left(mol\right)\\b=0,2\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}m_{C_2H_5OH}=0,8.46=36,8\left(g\right)\\m_{CH_3COOH}=0,2.60=12\left(g\right)\end{matrix}\right.\\ \Rightarrow\left\{{}\begin{matrix}\%m_{C_2H_5OH}=\dfrac{36,8}{48,8}.100\%=75,41\%\\\%m_{CH_3COOH}=100\%-75,41\%=24,59\%\end{matrix}\right.\)
b, PTHH:
\(C_2H_5OH+CH_3COOH\xrightarrow[t^o]{H_2SO_4đặc}CH_3COOC_2H_5+H_2O\)
LTL: 0,8 > 0,2 => Rượu dư
\(n_{CH_3COOC_2H_5\left(tt\right)}=0,2.85\%=0,17\left(mol\right)\\ m_{este}=0,17.88=14,96\left(g\right)\)
a.Gọi \(\left\{{}\begin{matrix}n_{C_2H_5OH}=x\\n_{CH_3COOH}=y\end{matrix}\right.\)
\(n_{H_2}=\dfrac{11,2}{22,4}=0,5mol\)
\(2C_2H_5OH+2Na\rightarrow2C_2H_5ONa+H_2\)
x 1/2 x ( mol )
\(2CH_3COOH+Na\rightarrow2CH_3COONa+H_2\)
y 1/2 y ( mol )
Ta có:
\(\left\{{}\begin{matrix}46x+60y=48,8\\\dfrac{1}{2}x+\dfrac{1}{2}y=0,5\end{matrix}\right.\) \(\Leftrightarrow\left\{{}\begin{matrix}x=0,8\\y=0,2\end{matrix}\right.\)
\(\rightarrow m_{C_2H_5OH}=0,8.46=36,8g\)
\(\rightarrow\left\{{}\begin{matrix}\%m_{C_2H_5OH}=\dfrac{36,8}{48,8}.100=75,4\%\\\%m_{CH_3COOH}=100\%-75,4\%=24,6\%\end{matrix}\right.\)
b.\(C_2H_5OH+CH_3COOH\rightarrow\left(H_2SO_4\left(đ\right),t^o\right)CH_3COOC_2H_5+H_2O\)
0,8 < 0,2 ( mol )
0,2 0,2 ( mol )
\(m_{CH_3COOC_2H_5}=0,2.88.85\%=14,96g\)

a) Gọi số mol CH3COOH, C2H5OH là a, b (mol)
=> 60a + 46b = 25,8 (1)
\(n_{H_2}=\dfrac{5,6}{22,4}=0,25\left(mol\right)\)
PTHH: 2Na + 2CH3COOH --> 2CH3COONa + H2
a------------------------->0,5a
2Na + 2C2H5OH --> 2C2H5ONa + H2
b--------------------->0,5b
=> 0,5a + 0,5b = 0,25 (2)
(1)(2) => a = 0,2 (mol); b = 0,3 (mol)
=> \(\left\{{}\begin{matrix}\%m_{CH_3COOH}=\dfrac{0,2.60}{25,8}.100\%=46,51\%\\\%m_{C_2H_5OH}=\dfrac{0,3.46}{25,8}.100\%=53,49\%\end{matrix}\right.\)
b)
\(n_{CH_3COOC_2H_5}=\dfrac{13,2}{88}=0,15\left(mol\right)\)
PTHH: CH3COOH + C2H5OH --H2SO4(đ),to--> CH3COOC2H5 + H2O
Xét tỉ lệ: \(\dfrac{0,2}{1}< \dfrac{0,3}{1}\) => Hiệu suất tính theo CH3COOH
PTHH: CH3COOH + C2H5OH --H2SO4(đ),to--> CH3COOC2H5 + H2O
0,15<---------------------------------0,15
=> \(H=\dfrac{0,15}{0,2}.100\%=75\%\)

\(n_{NaOH}=0,2.1=0,2\left(mol\right)\\ a,CH_3COOH+NaOH\rightarrow CH_3COONa+H_2O\\ n_{CH_3COOH}=n_{NaOH}=0,2\left(mol\right)\\ b,m_{CH_3COOH}=0,2.60=12\left(g\right)\\ m_{C_2H_5OH}=20-12=8\left(g\right)\)

\(a,n_{NaOH}=1,5.0,2=0,3\left(mol\right)\\ n_{H_2}=\dfrac{5,6}{22,4}=0,25\left(mol\right)\)
PTHH:
CH3COOH + NaOH ---> CH3COONa + H2O
0,3<-----------0,3
2CH3COOH + 2Na ---> 2CH3COONa + H2
0,3----------------------------------------------->0,15
2C2H5OH + 2Na ---> 2C2H5ONa + H2
0,2<---------------------------------------0,1
=> m = 0,2.46 +0,3.60 = 27,2 (g)
b) \(\left\{{}\begin{matrix}\%m_{CH_3COOH}=\dfrac{0,3.60}{27,2}.100\%=66,18\%\\\%m_{C_2H_5OH}=100\%-66,18\%=33,82\%\end{matrix}\right.\)

% khối lượng CH 3 COOH : 1,2/1,66 x 100% = 72,29%
% khối lương C 2 H 5 OH : 0,46/1,66 x 100% = 27,71%

a, \(CH_3COOH+Na\rightarrow CH_3COONa+\dfrac{1}{2}H_2\)
\(C_2H_5OH+Na\rightarrow C_2H_5ONa+\dfrac{1}{2}H_2\)
\(CH_3COOH+NaOH\rightarrow CH_3COONa+H_2O\)
b, Ta có: \(n_{NaOH}=0,2.0,5=0,1\left(mol\right)\)
Theo PT: \(n_{CH_3COOH}=n_{NaOH}=0,1\left(mol\right)\)
\(n_{H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
Theo PT: \(n_{H_2}=\dfrac{1}{2}n_{CH_3COOH}+\dfrac{1}{2}n_{C_2H_5OH}=0,3\)
\(\Rightarrow n_{C_2H_5OH}=0,5\left(mol\right)\)
\(\Rightarrow m=m_{CH_3COOH}+m_{C_2H_5OH}=0,1.60+0,5.46=29\left(g\right)\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{CH_3COOH}=\dfrac{0,1.60}{29}.100\%\approx20,69\%\\\%m_{C_2H_5OH}\approx79,31\%\end{matrix}\right.\)

a)
- Xét TN2:
nNaOH = 0,2.0,2 = 0,04 (mol)
PTHH: CH3COOH + NaOH --> CH3COONa + H2O
0,04<-----0,04
- Xét TN1:
\(n_{H_2}=\dfrac{0,672}{22,4}=0,03\left(mol\right)\)
PTHH: 2CH3COOH + 2Na --> 2CH3COONa + H2
0,04------------------------------>0,02
2C2H5OH + 2Na --> 2C2H5ONa + H2
0,02<--------------------------0,01
=> m = 0,04.60 + 0,02.46 = 3,32 (g)
b) \(\left\{{}\begin{matrix}\%m_{CH_3COOH}=\dfrac{0,04.60}{3,32}.100\%=72,29\%\\\%m_{C_2H_5OH}=\dfrac{0,02.46}{3,32}.100\%=27,71\%\end{matrix}\right.\)


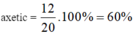


\(n_{NaOH}=0,4.0,5=0,2\left(mol\right)\)
PT: \(CH_3COOH+NaOH\rightarrow CH_3COONa+H_2O\)
Theo PT: \(n_{CH_3COOH}=n_{NaOH}=0,2\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{CH_3COOH}=\dfrac{0,2.60}{15,2}.100\%\approx78,95\%\\\%m_{C_2H_5OH}\approx21,05\%\end{matrix}\right.\)