Hòa tan 13g hỗn hợp gồm AL và Fe vừa đủ trong 150ml dung dịch HCL thu được 6,72 lit khí (đkc) a. Tính phần trăm khối lượng mỗi chất trong hỗn hợp b. Tính nồng độ mol dung dịch HCL c. Tính khối lượng muối sinh ra
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


a) Gọi $n_{Al} = a(mol) ; n_{Fe} = b(mol) \Rightarrow 27a + 56b = 33,4(1)$
$2Al + 6HCl \to 2AlCl_3 + 3H_2$
$Fe + 2HCl \to FeCl_2 + H_2$
Theo PTHH : $n_{H_2} = 1,5a + b = \dfrac{17,92}{22,4} = 0,8(2)$
Từ (1)(2) suy ra : a = 0,2 ; b = 0,5
$\%m_{Al} = \dfrac{0,2.27}{33,4}.100\% = 16,17\%$
$\%m_{Fe} = 100\% - 16,17\% = 83,83\%$
b) $n_{HCl} = 2n_{H_2} = 1,6(mol)$
c) $m_{muối} = m_{hh} + m_{HCl} - m_{H_2} = 33,4 + 1,6.36,5 - 0,8.2 = 90,2(gam)$

a, Ta có: 27nAl + 56nFe = 22 (1)
PT: \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
Theo PT: \(n_{H_2}=\dfrac{3}{2}n_{Al}+n_{Fe}=\dfrac{19,832}{24,79}=0,8\left(mol\right)\left(2\right)\)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}m_{Al}=0,4\left(mol\right)\\n_{Fe}=0,2\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{Al}=\dfrac{0,4.27}{22}.100\%\approx49,09\%\\\%m_{Fe}\approx50,91\%\end{matrix}\right.\)
b, \(n_{HCl}=2n_{H_2}=1,6\left(mol\right)\)
\(\Rightarrow C_{M_{HCl}}=\dfrac{1,6}{0,5}=3,2\left(M\right)\)

\(n_{HCl}=0,3.2=0,6\left(mol\right)\\ n_{H_2}=\dfrac{5,6}{22,4}=0,25\left(mol\right)\\ 2Al+6HCl\rightarrow2AlCl_3+3H_2\\ Fe+2HCl\rightarrow FeCl_2+H_2\\ Vì:\dfrac{0,6}{2}>\dfrac{0,25}{1}\Rightarrow HCldư\\ Đặt:n_{Al}=t\left(mol\right);n_{Fe}=r\left(mol\right)\\ \left(t,r>0\right)\\ \Rightarrow\left\{{}\begin{matrix}27t+56r=8,3\\1,5t+r=0,25\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}t=0,1\\r=0,1\end{matrix}\right.\\ \Rightarrow m_{Al}=0,1.27=2,7\left(g\right);m_{Fe}=0,1.56=5,6\left(g\right)\\ b,n_{AlCl_3}=n_{Al}=0,1\left(mol\right)\Rightarrow m_{AlCl_3}=0,1.133,5=13,35\left(g\right)\\ n_{Fe}=n_{FeCl_2}=0,1\left(mol\right)\Rightarrow m_{ddFeCl_2}=127.0,1=12,7\left(g\right)\\ m_{ddHCl}=300.1,15=345\left(g\right)\\ m_{ddsau}=8,3+345-0,25.2=352,8\left(g\right)\)
\(n_{HCl\left(dư\right)}=0,6-0,25.2=0,1\left(mol\right)\\ \Rightarrow m_{ddHCl}=0,1.36,5=3,65\left(g\right)\\ C\%_{ddHCl\left(dư\right)}=\dfrac{3,65}{352,8}.100\approx1,035\%\\ C\%_{ddAlCl_3}=\dfrac{13,35}{352,8}.100\approx3,784\%\\ C\%_{ddFeCl_2}=\dfrac{12,7}{352,8}.100\approx3,6\%\)

Gọi \(\left\{{}\begin{matrix}n_{Al}=x\left(mol\right)\\n_{Fe}=y\left(mol\right)\end{matrix}\right.\)
\(n_{H_2}=\dfrac{4,48}{22,4}=0,2mol\)
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
Theo pt: \(\Rightarrow\left\{{}\begin{matrix}3x+y=0,2\\27x+56y=5,5\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}x=\dfrac{19}{470}\\y=\dfrac{37}{470}\end{matrix}\right.\)
\(\%m_{Al}=\dfrac{\dfrac{19}{470}\cdot27}{5,5}\cdot100\%=19,84\%\)
\(\%m_{Fe}=100\%-19,84\%=80,16\%\)

a, Ta có: 27nAl + 56nFe = 27,8 (1)
PT: \(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\)
\(Fe+H_2SO_4\rightarrow FeSO_4+H_2\)
Theo PT: \(n_{H_2}=\dfrac{3}{2}n_{Al}+n_{Fe}=\dfrac{17,353}{24,79}=0,7\left(mol\right)\left(2\right)\)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}n_{Al}=0,2\left(mol\right)\\n_{Fe}=0,4\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{Al}=\dfrac{0,2.27}{27,8}.100\%\approx19,42\%\\\%m_{Fe}\approx80,58\%\end{matrix}\right.\)
b, \(n_{H_2SO_4}=n_{H_2}=0,7\left(mol\right)\Rightarrow C_{M_{H_2SO_4}}=\dfrac{0,7}{0,5}=1,4\left(M\right)\)

a,Fe + 2HCl → FeCl + H2 (1)
FeO + 2HCl → FeCl + H2O (2)
nH2 = 3,36/ 22,4 = 0,15 ( mol)
Theo (1) nH2 = nFe = 0,15 ( mol)
mFe = 0,15 x 56 = 8.4 (g)
m FeO = 12 - 8,4 = 3,6 (g)
a, \(n_{H_2}=\frac{3,36}{22,4}=0,15\left(mol\right)\)
\(Fe+2HCl->FeCl_2+H_2\left(1\right)\)
\(FeO+2HCl->FeCl_2+H_2O\left(2\right)\)
theo (1) \(n_{Fe}=n_{H_2}=0,15\left(mol\right)\)
=> \(m_{Fe}=0,15.56=8,4\left(g\right)\)
=> \(m_{FeO}=12-8,4=3,6\left(g\right)\)

ta thấy : nFe =nH2 = 0,15
=> mFe =0,15 x 56 = 8,4g
%Fe=8,4/12 x 100 = 70%
=>%FeO = 100 - 70 = 30%
b) BTKLra mdd tìm mct of HCl
c) tìm mdd sau pứ -mH2 nha bạn

a) PTHH: \(Fe+2HCl\rightarrow FeCl_2+H_2\uparrow\)
a_____2a______a_____a (mol)
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\uparrow\)
b_____3b_______b_____\(\dfrac{3}{2}\)b (mol)
Ta lập HPT: \(\left\{{}\begin{matrix}56a+27b=36,1\\a+\dfrac{3}{2}b=\dfrac{21,28}{22,4}=0,95\end{matrix}\right.\) \(\Leftrightarrow\left\{{}\begin{matrix}a=0,5\\b=0,3\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}m_{Fe}=0,5\cdot56=28\left(g\right)\\m_{Al}=8,1\left(g\right)\end{matrix}\right.\)
b+c) Theo các PTHH: \(\left\{{}\begin{matrix}n_{HCl}=2a+3b=1,9\left(mol\right)\\n_{FeCl_2}=0,5\left(mol\right)\\n_{AlCl_3}=0,3\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}C_{M_{HCl}}=\dfrac{1,9}{0,2}=9,5\left(M\right)\\C_{M_{FeCl_2}}=\dfrac{0,5}{0,2}=2,5\left(M\right)\\C_{M_{AlCl_3}}=\dfrac{0,3}{0,2}=1,5\left(M\right)\end{matrix}\right.\)


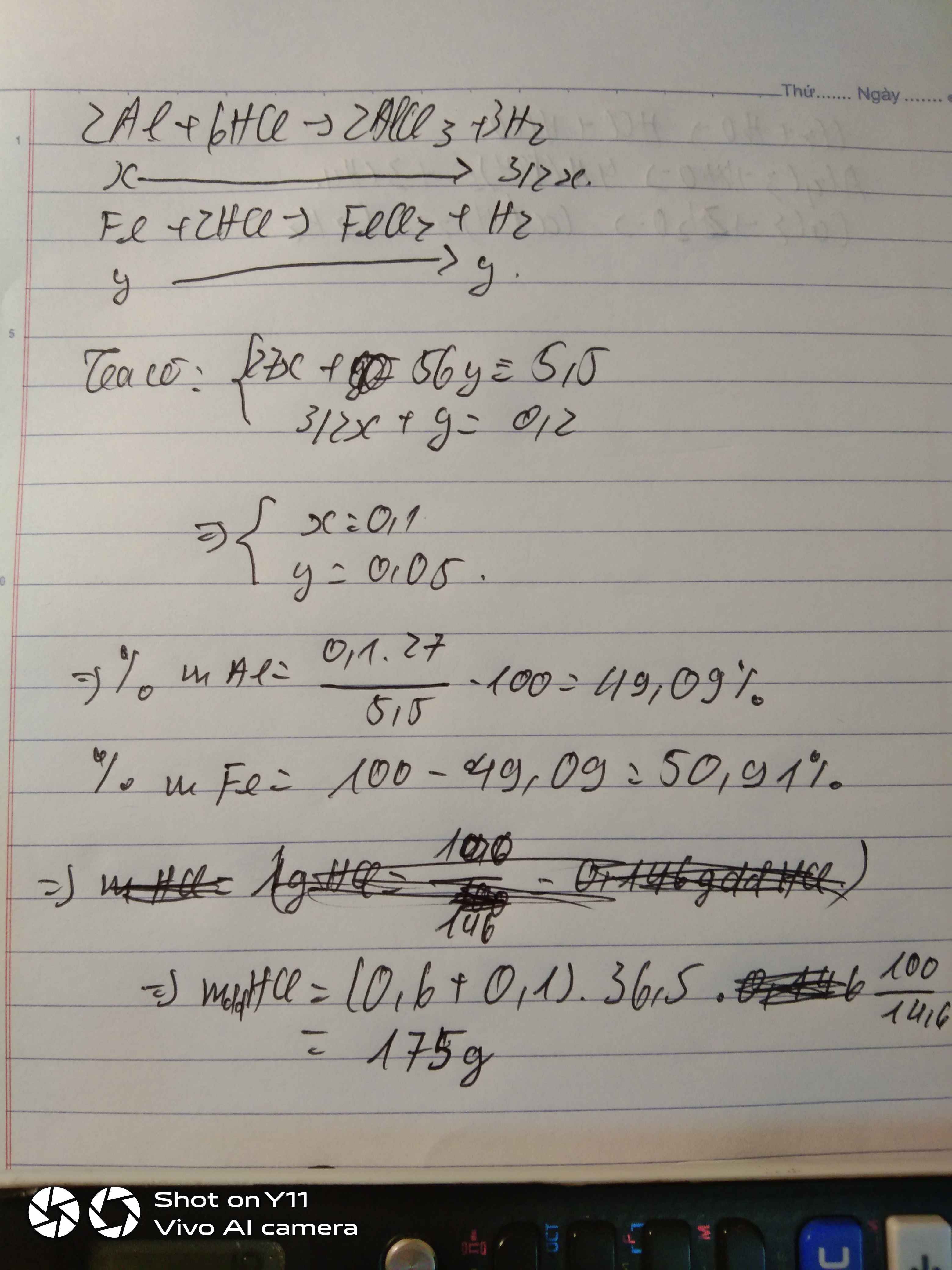

a) Gọi $n_{Al} =a (mol) ; n_{Fe} = b(mol) \Rightarrow 27a + 56b = 13(1)$
$2Al + 6HCl \to 2AlCl_3 + 3H_2$
$Fe + 2HCl \to FeCl_2 + H_2$
Theo PTHH : $n_{H_2} = 1,5a + b = \dfrac{6,72}{22,4} = 0,3(2)$
Từ (1)(2) suy ra : $a = \dfrac{1}{15} ; b = 0,2$
$\%m_{Al} = \dfrac{ \dfrac{1}{15}.27}{13}.100\% = 13,8\%$
$\%m_{Fe} = 100\% - 13,8\% = 86,2\%$
b) $n_{HCl} = 2n_{H_2} = 0,3.2 = 0,6(mol)$
$\Rightarrow C_{M_{HCl}} = \dfrac{0,6}{0,15} = 4M$
c) $n_{muối} = m_{kim\ loại} + m_{HCl} - m_{H_2} = 13 + 0,6.36,5 - 0,3.2 = 34,3(gam)$