Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a. PTHH: Fe + 2HCl ---> FeCl2 + H2 (1)
b, \(n_{Fe}=\dfrac{11,2}{56}=0,2\left(mol\right)\)
Theo pthh (1): \(n_{H_2}=n_{Fe}=0,2\left(mol\right)\)
\(\rightarrow V_{H_2}=0,2.22,4=4,48\left(l\right)\)
c, PTHH: 2H2 + O2 --to--> 2H2O (2)
Theo pthh (2): \(n_{O_2}=\dfrac{1}{2}n_{H_2}=\dfrac{1}{2}.0,2=0,1\left(mol\right)\)
\(\rightarrow m_{O_2}=0,1.32=3,2\left(g\right)\)

1/
\(CaCO_3 + 2HCl \to CaCl_2 + CO_2 +H_2O\\ CO_2 + NaOH \to NaHCO_3 2NaHCO_3 \xrightarrow{t^o} Na_2CO_3 + CO_2 + H_2O\\ Na_2CO_3 + BaCl_2 \to BaCO_3 + 2NaCl\)
1)
\(CaCO_3\underrightarrow{t^o}CO_2+CaO\\ NaOH+CO_2\rightarrow NaHCO_3\\ NaHCO_3+NaOH\rightarrow Na_2CO_3+H_2O\\ Na_2CO_3+Ba\left(OH\right)_2\rightarrow NaOH+BaCO_3\)
2)
\(n_{HCl}=C_{M_{HCl}}.V_{HCl}=1.0,2=0,2\left(mol\right)\)
PTHH: \(K_2CO_3+2HCl\rightarrow2KCl+H_2O+CO_2\)
\(\Rightarrow n_{K_2CO_3}=n_{CO_2}=0,1\left(mol\right)\)
a) \(V_{CO_2\left(đktc\right)}=0,1.22,4=2,24\left(l\right)\)
b) \(m_{K_2CO_3}=0,1.138=13,8\left(g\right)\)
\(m_{ddK_2CO_3}=\dfrac{13,8.100}{13,8}=100\left(g\right)\)

nZn= 13/65=0,2(mol)
a) PTHH: Zn + 2 HCl -> ZnCl2 + H2
b) nH2=nZnCl2=nZn=0,2(mol)
=>V(H2,đktc)=0,2 x 22,4= 4,48(l)
c) khối lượng muối sau phản ứng chứ nhỉ?
mZnCl2=136.0,2=27,2(g)

\(n_{Zn}=\dfrac{6,5}{65}=0,1mol\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
0,1 0,2 0,1 0,1
\(V_{H_2}=0,1\cdot22,4=2,24l\)

a, \(Zn+2HCl\rightarrow ZnCl_2+H_2\)
b, \(n_{Zn}=\dfrac{39}{65}=0,6\left(mol\right)\)
Theo PT: \(n_{HCl}=2n_{Zn}=1,2\left(mol\right)\Rightarrow m_{HCl}=1,2.36,5=43,8\left(g\right)\)
c, \(n_{H_2}=n_{Zn}=0,6\left(mol\right)\Rightarrow V_{H_2}=0,6.24,79=14,874\left(l\right)\)
d, - Quỳ tím hóa đỏ do HCl dư.

a.b.\(n_{Zn}=\dfrac{1,95}{65}=0,03mol\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
0,03 0,06 0,03 0,03 ( mol )
\(V_{H_2}=0,03.22,4=0,672l\)
\(m_{ddHCl}=\dfrac{0,06.36,5}{7,3\%}=30g\)
c.Tên muối: Kẽm clorua
\(m_{ZnCl_2}=0,03.136=4,08g\)
\(m_{ddspứ}=30+1,95-0,03.2=31,89g\)
\(C\%_{ZnCl_2}=\dfrac{4,08}{31,89}.100\%=12,79\%\)

a) \(PTHH:Mg+2HCl\rightarrow MgCl_2+H_2\)
b) \(n_{Mg}=\dfrac{4,8}{24}=0,2mol\)
\(n_{HCl}=2.n_{Mg}=0,2.2=0,4mol\)
\(\Rightarrow m_{HCl}=n.M=0,4.36,5=14,6g\)
c) \(n_{H_2}=n_{Mg}=0,2mol\)
Thể tích khí hidro sinh ra (ở đktc):
\(V_{H_2}=0,2.24,79=4,958l.\)

\(n_{Zn}=\dfrac{13}{65}=0,2\left(mol\right)\\
pthh:Zn+2HCl\rightarrow ZnCl_2+H_2\)
0,2 0,4 0,2
\(V_{H_2}=0,2.22,4=4,48\left(l\right)\\
C\%_{HCl}=\dfrac{0,4.36,5}{200}.100\%=7,3\%\)
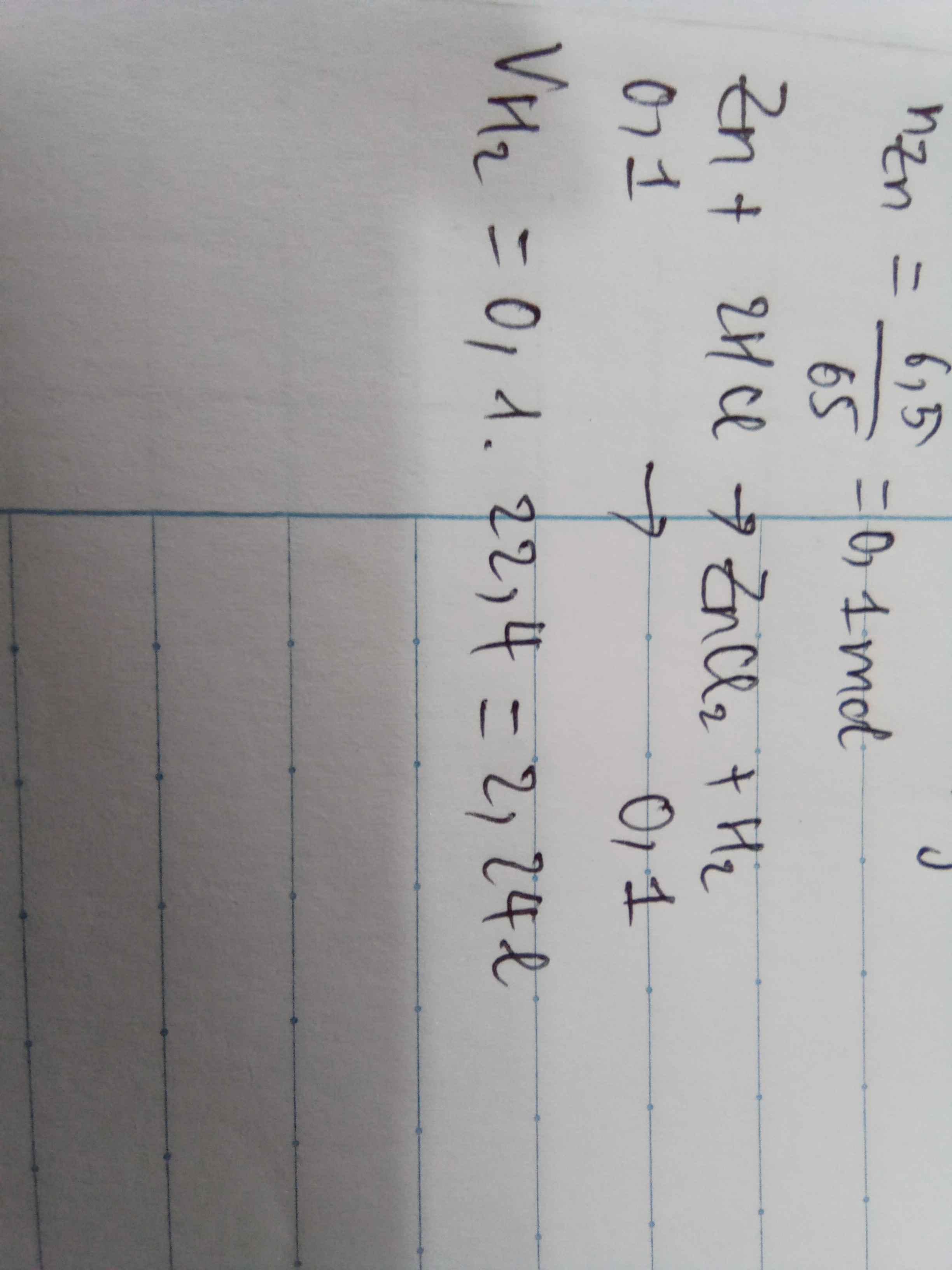
nZn = 19,5/65 = 0,3 (mol)
PTHH: Zn + 2HCl -> ZnCl2 + H2
Mol: 0,3 ---> 0,6 ---> 0,3 ---> 0,3
VH2 = 0,3 . 22,4 = 6,72 (l)
mHCl = 0,6 . 36,5 = 21,9 (g)
\(n_{Zn}=\dfrac{19,5}{65}=0,3mol\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
0,3 0,6 0,3 ( mol )
\(V_{H_2}=0,3.22,4=6,72l\)
\(m_{HCl}=0,6.36,5=21,9g\)